FDA won't comment on status of Emergency Use Authorizations for two antibody treatments
Por um escritor misterioso
Descrição
The US Food and Drug Administration told CNN Thursday morning that the agency doesn’t have any comments on the applications for Emergency Use Authorizations for Eli Lilly and Regeneron antibody treatments.
The coronavirus pandemic has brought countries to a standstill. In many places, as countries reopen, Covid-19 cases are on the rise. Follow here for the latest.
The coronavirus pandemic has brought countries to a standstill. In many places, as countries reopen, Covid-19 cases are on the rise. Follow here for the latest.

Moderna asks the F.D.A. to authorize its vaccine for children

Not Ready for the End Game — Why Ending Federal Covid-19 Emergency

Federal Register :: Authorizations of Emergency Use of Certain

Evusheld to prevent Covid-19: There won't be nearly enough for

Federal Register :: Authorizations of Emergency Use of Certain
FDA Open Meeting on Johnson & Johnson COVID-19 Vaccine, Part 5

Regulatory tracker: EMA backs Vertex's gene-editing therapy

Doctor Submits Fluvoxamine EUA Application to FDA
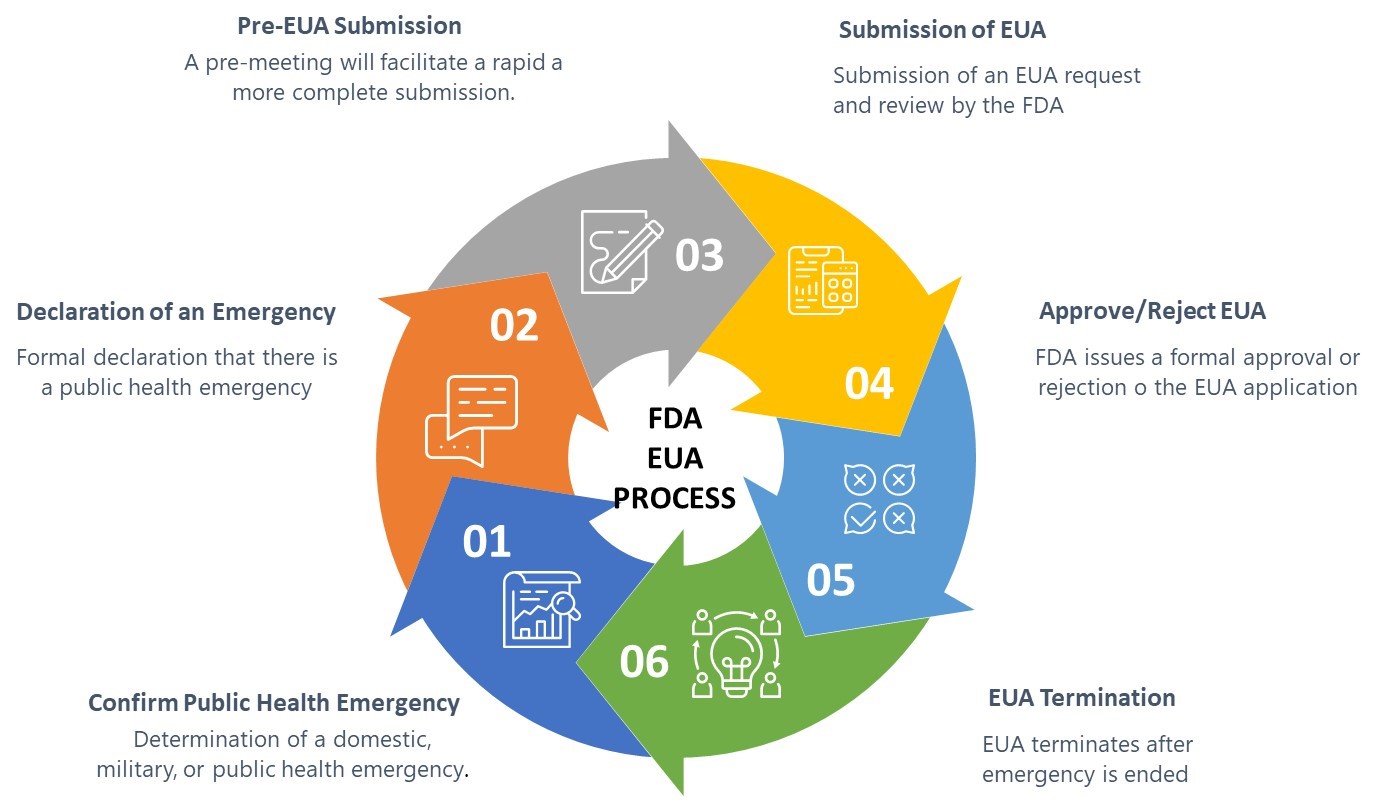
Navigating The FDA's Emergency Use Authorization Process

FDA pulls authorization for COVID antibody treatment over lack of
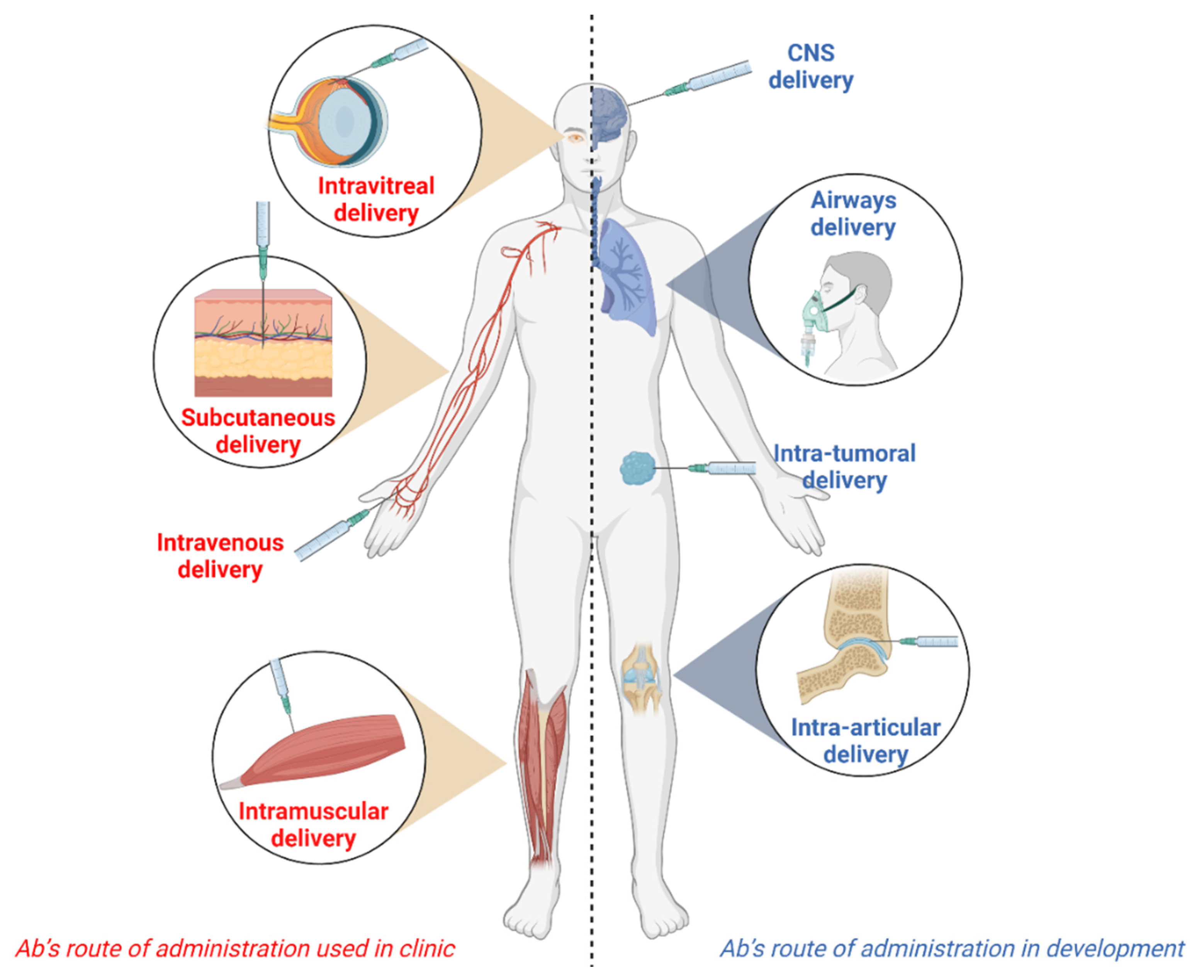
Antibodies, Free Full-Text

Federal Register :: Authorizations of Emergency Use of Certain

IJMS, Free Full-Text
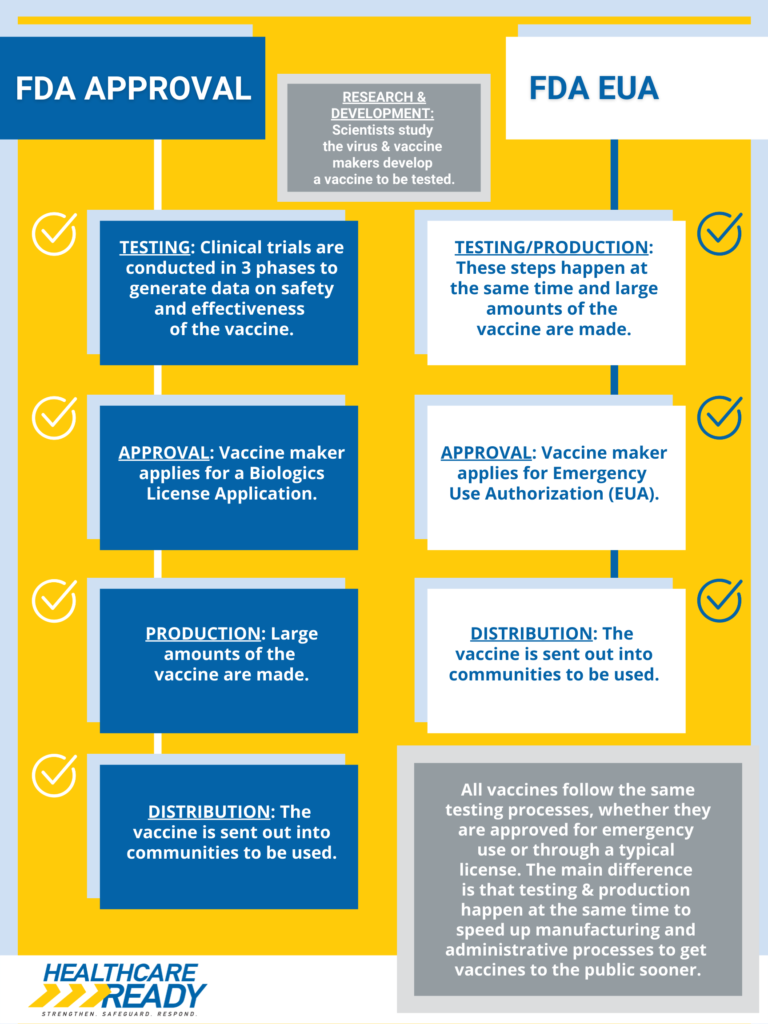
Infographic: FDA Approval vs. FDA Emergency Use Authorization
de
por adulto (o preço varia de acordo com o tamanho do grupo)






